December 13, 2017

Lexington, Concord and Medicaid Drug Formularies: Massachusetts Fires Another “Shot Heard Round the World”
Market Corner Commentary for December 13, 2017
On the 19th of April in 1775, a ragtag Massachusetts militia confronted the mighty British army in search of Colonial munitions and rebellion leaders. The Battle of Lexington and Concord marked the first military engagement between American revolutionaries and their British colonizers. The Americans carried the day and began their long arduous march toward independence.
Poet Ralph Waldo Emerson captured the battle’s mythical meaning to American patriots at the July 4, 1837 dedication ceremony for the Battle of Concord memorial. In his “Concord Hymn” Emerson wrote:
Here once the embattled farmers stood,
And fired the shot heard round the world.
Patriot battle fervor has now returned to Massachusetts. Modern-day revolutionaries have fired “a shot” at the pharmaceutical industry through a broad CMS waiver request. It includes provisions that would enable Massachusetts to manage Medicaid drug formularies like commercial enterprises.
The repercussions of this Massachusetts waiver request are being “heard round the Medicaid world.” If granted, Massachusetts and other like-minded states will gain the freedom to design their Medicaid programs in ways that enhance care outcomes and lower costs.
Like the British army, the pharmaceutical industry will wage a furious battle to maintain an unacceptable status quo. Healthcare “patriots” must stand with Massachusetts and fight for better, more holistic and more equitable healthcare services.

Breaking Free
On September 8th, the Massachusetts Office of Health and Human Services filed a Section 1115 Amendment Request to the Centers for Medicare and Medicaid Services (CMS). The request incorporates provisions that would enable Massachusetts Medicaid to negotiate drug formularies like commercial insurers and pharmacy benefit managers (PBMs).
Specifically, Massachusetts Medicaid wants CMS to grant it the authority to implement the following new policies:[1]
- Adopt closed formularies with at least one drug available for each therapeutic class (i.e., disease)
- Exclude drugs from formularies “with limited or inadequate evidence of clinical efficacy”
These provisions may seem “wonky” but they strike at the heart of Big Pharma’s ability to force state Medicaid programs to pay sky-high prices for branded drugs. Current law requires Medicaid to offer beneficiaries all branded drugs for which pharma manufacturers provide discounts.
Adopting closed formularies for a limited number of drugs will allow MassHealth to negotiate better discounts based on larger volumes for preferred drugs. The proposed waiver would include processes for patients to receive alternate drugs when medically necessary.
MassHealth is also asking for the ability to decide whether or not to cover drugs that have not yet been proven clinically effective. Commercial insurers already enjoy this discretion.
Here are the freedoms for which MassHealth is fighting:
Freedom from Open Formularies
Revenue rules. Even with percentage discounts, drug manufacturers can raise prices to optimize revenue from sales to Medicaid programs. The following chart details the considerable revenue that pharma manufacturers generate by selling their blockbuster drugs to Medicaid programs.
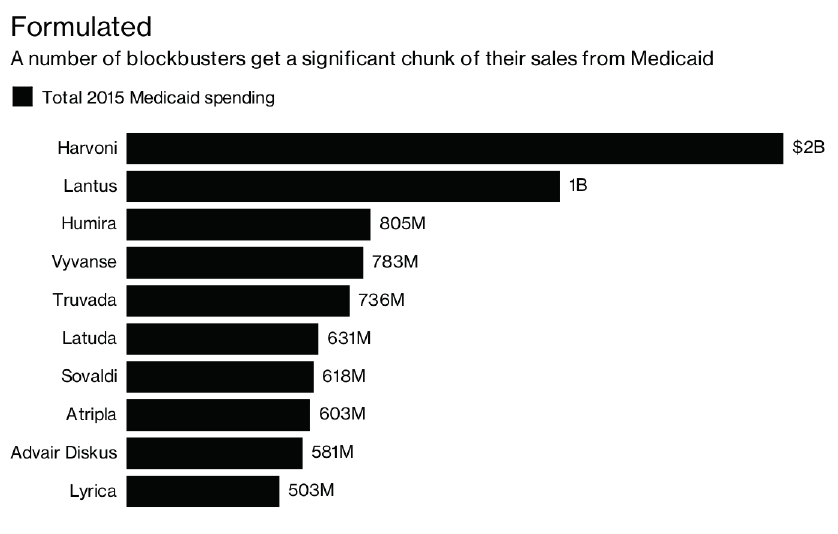
Source: Bloomberg
In contrast, commercial PBMs, as well as traditional Medicare and Medicare Advantage Rx plans, exclude higher-cost drugs from their formularies when there are effective lower-cost alternatives. In response, drug manufacturers offer substantial rebates to incentivize PBMs to include their drugs within specific disease formularies.
With rising drug prices, PBMs are more aggressively excluding high-cost drugs from their formularies. The following chart shows that the nation’s two largest PBMs, CVS and Express Scripts, have more than doubled the number of excluded drugs since 2014.
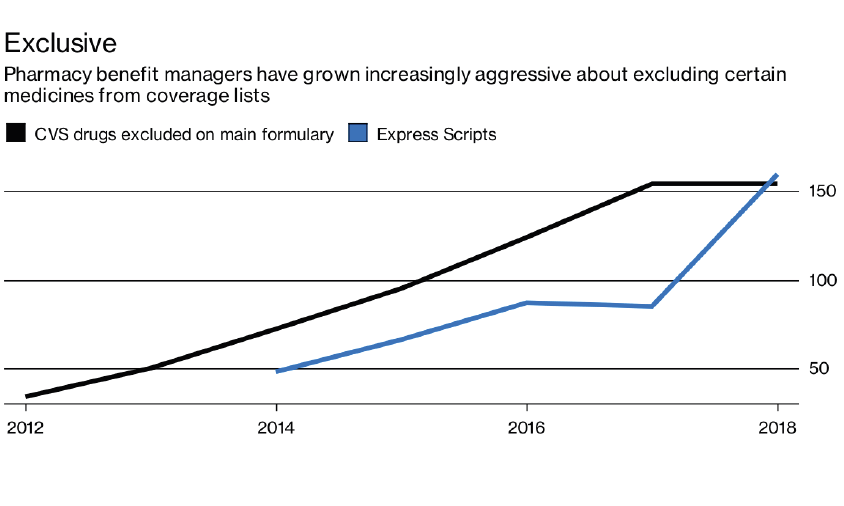
Source: Bloomberg
Freedom from Marginal Drugs
Enacted in December 2016, the 21st Century Cures Act grants expedited Food and Drug Administration (FDA) review and approval for drugs that “treat serious conditions and fill an unmet medical need.” 30 million Americans suffer from one of roughly 7,000 rare “orphan” diseases.[2] Expedited approvals mean drugs can come to market without definitive evidence of their efficacy.
Pharmaceutical companies are actively developing drugs that treat these rare diseases because they’re able to charge very high prices for drugs that gain FDA approval. Under current law, the FDA does not incorporate a drug’s price into its approval process. As a consequence, FDA approval can translate into monopolistic pricing power for drug manufacturers.
For example, Biogen and Sarepta Therapeutics received FDA approval last December for drugs that treat rare forms of muscular dystrophy. Clinical trials for these drugs have been promising, but not conclusive. Commercial insurers, such as Anthem and United HealthCare, have resisted approving the drugs for widespread use because of their whopping price tags and their limited evidence of efficacy.
Biogen’s Spinraza costs $750,000 the first year and $375,000 per year afterward. Patients must take the drug for life. Spinraza will not cure spinal muscular atrophy (SMA), but has demonstrated some efficacy in trials with very young children. The FDA has approved Spinraza for use with all SMA patients irrespective of age.[3]
Unlike commercial insurers and even Medicare, state Medicaid programs almost always pay for unproven and/or marginally effective high-cost drugs like Spinraza. If CMS approves their waiver request, Massachusetts Medicaid will have the administrative authority to decide when and when not to cover high-cost drugs for “orphan” diseases. Patients would be able to appeal non-coverage decisions.

With the Eyes of All People Upon Us
Eleven ships carrying over a thousand English Puritans established the Massachusetts Bay Colony in 1630. Just before landfall, future governor John Winthrop gave a stirring sermon on the Arbella and presented his vision for the new colony:[4]
We shall be as a city upon a hill, the eyes of all people are upon us.
John Winthrop’s now-famous Massachusetts sermon is the first evidence of a unique and powerful American mindset. Modern politicians from John Kennedy to Ronald Reagan to Barack Obama to George W. Bush to Mitt Romney have employed Winthrop’s “city upon a hill” metaphor to reference American exceptionalism.
While American exceptionalism conquered a continent, won two world wars and created an economic innovation engine second to none, it has also created an inefficient, fragmented health care system.
In 2006 under then-Governor Romney, Massachusetts instituted market-based healthcare reforms that became the legislative model for Obamacare. Today, Massachusetts is taking a leading role in reforming Medicaid through its waiver request.
All over America, state Medicaid directors are watching and waiting with riveted interest to see whether CMS grants Massachusetts its desired reforms. If they do, other states will flood CMS with similar requests for administrative freedom. In the process, Medicaid will strike a powerful blow for more rational healthcare delivery.
Big Pharma and their supporters are fighting back, noting that low-income Medicaid patients and the elderly will disproportionately suffer if Medicaid programs deny payment for promising medications.[5] Advocacy groups for specific diseases place enormous pressure on commercial and governmental insurers to pay for expensive drugs.
The financial pressure on state Medicaid programs is crushing. Medicaid is the largest and fastest growing component of Massachusetts’ state budget. It currently represents nearly 40% of total state expenditures, including the federal government’s contribution.[6]
Within Massachusetts Medicaid, prescription drug costs more than doubled between 2010 ($917 million) and 2016 ($1.94 billion) with no relief in sight. This translates into a 13% compounded annual growth rate that threatens to “crowd out” other vital social and health care spending.
Paraphrasing Shakespeare’s Hamlet, “there’s the rub.” American healthcare spends astronomical sums on marginal pharmaceutical treatments and unnecessary acute care services. At the same time, the system woefully underfunds vital behavioral health, chronic disease management, and preventive and social care services that advance health and wellness.
President Trump has demanded that Big Pharma address “outrageous drug prices” and has promoted greater state control over Medicaid program design. Massachusetts’ Section 1115 Amendment request to CMS gives the Trump administration an opportunity to turn the President’s rhetoric into policies that benefit the American people.

Standing Up for Better Healthcare
In long-ago April 1776, brave Americans in Massachusetts took a stand against British tyranny. They had no knowledge their actions would trigger the Declaration of Independence, a long bloody war and the birth of a new nation. Against all odds, they decided to fight and perhaps die for a better future.
As a nation, America already spends more than enough money on healthcare. “We the People” deserve better. U.S. healthcare confronts a “distribution” not a “funding” challenge. The system needs to spend its dollars more wisely.
A good place to start is by granting Medicaid programs the same autonomy the law grants private enterprise to manage pharmaceutical care. American healthcare will become great when it acknowledges that outcomes matter, customers count
[1] http://www.medicaidandthelaw.com/2017/09/13/baker-administration-submits-1115-waiver-request-to-cms-including-major-change-to-drug-coverage/
[2] https://www.nytimes.com/2017/06/22/health/duchenne-muscular-dystrophy-drug-exondys-51.html
[3] https://www.nytimes.com/2016/12/30/business/spinraza-price.html?_r=1
[4] http://www.ushistory.org/us/3c.asp
[5] https://www.forbes.com/sites/dougschoen/2017/09/26/massachusetts-dangerous-waiver-program-a-poor-response-to-nations-crumbling-health-care-system/#3e82fbb81339
[6] http://massbudget.org/report_window.php?loc=What-Is-the-Actual-State-Cost-of-MassHealth-in-2018.html





