June 4, 2020
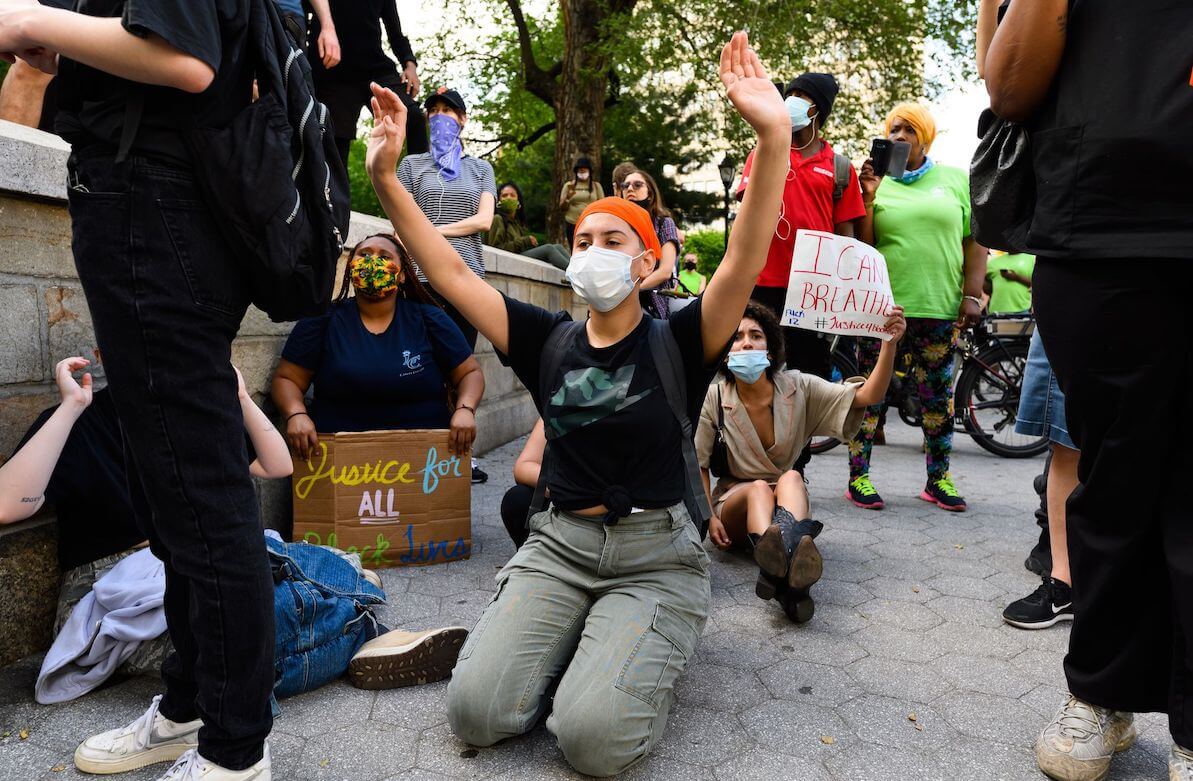
Rogue Cops and Vigilantes: COVID-19’s Best Friends
Key Takeaways
- Widespread civil unrest in the wake of the police and vigilante murders of two African Americans has set back the effort to control the COVID-19 pandemic.
- Public health officials continue to believe far more extensive testing, tracing and, if necessary, quarantining of sick and exposed individuals will be necessary to end the pandemic and reopen the economy.
- Testing sites in many cities closed over concerns about violence during civil unrest because of ongoing demonstrations in dozens of American cities and towns.
- The federal government continues to play almost no role in promoting testing-and-tracing.
- Cash-strapped state and local officials will require additional resources to implement tracing programs, which Congress has yet to consider.
In Albert Camus’ 1947 classic The Plague, people in the Algerian town of Oran are dying in isolated pockets from a mysterious ailment that looks suspiciously like bubonic plague. Local physicians warn the town’s top official that half the population will be wiped out unless he orders an immediate quarantine. He insists the doctors declare the outbreak is in fact the plague before he acts.
They demure. They cannot state that yet with scientific certainty.
“Your view, I take it,” the Prefect puts in, “is this. Even if it isn’t plague, the prophylactic measures enjoined by law for coping with a state of plague should be put in force immediately?”
“If you insist in my having ‘a view,’ that conveys it accurately enough,” the town’s chief physician responds.
The Federal Government’s Refusal to Take Responsibility
With the body count from COVID-19 now beyond 105,000, America’s political and medical leaders are responding like Oran’s. The federal government is refusing to take responsibility for coordinating a nationwide program that all responsible scientific officials recommend: systematic testing to identify everyone infected; systematic tracing of everyone that’s been in contact with those sick people; and quarantining of exposed individuals when necessary.
State and local officials, meanwhile, are being buffeted between their understandable desire to reopen their economies and their public health departments’ advice for how to combat the disease. They have vacillated between stern warnings and contradictory half measures that have little chance of bringing closure to the pandemic before there’s a vaccine.
And now, on top of this world-worst response to the first two months of the pandemic, the murders of Ahmaud Arbery and George Floyd, both African-Americans, by self-appointed vigilantes and a white Minneapolis policeman, have set off massive protests that will inevitably contribute to spreading the disease. The White House’s inflammatory rhetorical response to those demonstrations heightens the probability that the pandemic will remain uncontrolled and politicized through the November election.
Reeling From an Unprecedented Triple Whammy
The U.S. is reeling from a simultaneous triple whammy, unprecedented in its history: the outbreak of an uncontrolled pandemic that takes a disproportionate toll on black, brown and poor Americans; an economic depression in a society already riven by vast disparities in wealth and income; and widespread civic unrest triggered by a nation’s inability to come to grips with its long-standing racial divisions. This is nothing less than a public health catastrophe.
The terrible events of late May and early June have dealt a devastating setback to local efforts to implement a comprehensive COVID-19 testing-and-tracing regime, which is key to a successful economic reopening. Dozens of mobile testing sites in California, Florida, Pennsylvania and Illinois have shut down temporarily. Walgreens and CVS report closing nearly two dozen of their testing sites.
This comes on top of the early stumble and delayed rollout in COVID-19 testing. In most areas of the country, testing remains well below the level epidemiologists and health experts say is necessary to control the pandemic. Earlier projections, made before the civil unrest broke out, showed that the U.S. would not reach necessary testing levels until mid-July. At the earliest.
States cannot reopen their economies safely without fast, accurate testing of people who have been exposed or are great risk of contracting COVID-19. It’s the first step mentioned in the restriction relaxation guidelines issued in mid-April by both the Centers for Disease Control and Prevention and the Infectious Disease Society of America. Fast results from comprehensive testing allow public health officials to immediately identify whether social distancing measures effectively limit the spread of the disease.
Testing Alone Is Not Sufficient
Testing alone is not sufficient. Isolation of people who test positive must follow. Then tracers must reach out to everyone contacted by that infected person over the prior three or four days – the average time it takes to begin showing symptoms.
This wider group must undergo testing, understand the benefits of aggressive social distancing, and isolate if necessary. This tracing activity will require a small army of trained outreach workers, only a small fraction of whom are already at work at state and local public health agencies across the U.S.
“We’re going to need 4 to 5 million tests per week, but we’re doing less than 2 million,” Dr. Anand Parekh, chief medical advisor at the Bipartisan Policy Center in Washington, D.C., said on May 7. As of early June, the U.S. is performing only 400,000 tests per day, which is 3 million a week.
“We need 300,000 contact tracers based on international standards,” he added. Others say that number could be as low as 100,000. Still, as of mid-May, the U.S. had less than half the minimum number.
The U.S. clearly has more work to do before most areas can reopen safely. Yet many states, given final authority for lifting restrictions by the Trump administration, have already relaxed social distancing. Some heavily populated states, like Texas, Georgia and Florida, are still at the front end of their outbreaks. Others are sparsely populated but have been hit by large outbreaks in nursing homes and large industrial facilities including meatpacking plants.
Those are the areas starting to show major upticks in their outbreaks. “We’re very much not out of the woods yet,” said Dr. Jennifer Nuzzo of Johns Hopkins Bloomberg School of Public Health said on June 2. “There’s at least 16 states that have shown rising caseloads each day for the past two weeks.”

Given the extent of interstate commerce and travel in the U.S., those states’ premature relaxation of prophylactic measures will make more rigorous efforts in other states less effective. It will slow everyone’s return to what some now call the new normal. Without an effective, approved, and widely distributed vaccine, some limits on social and economic gathering will remain in many areas of the country. The first quarter of 2021 is the earliest anyone projects for that possibility.
Deep Political Divisions Weaken Recovery
The U.S.’s inability to commit to the comprehensive strategy recommended by public health and medical professionals reflects the nation’s deep political divisions. Those long preceded the pandemic. President Donald Trump’s frequent waffling on the first principle of the strategy – testing – has only served to widen the divide.
“Anybody right now and yesterday, anybody that needs a test, they’re there. And the tests are beautiful,” Trump said at the CDC in Atlanta on March 6. None of those assertions were true then, just as they’re not true now. With testing finally ramping up, he recently asserted that “it could be that testing is, frankly, overrated. … When you test, you have a case. When you test, you find something is wrong with people. If we didn’t do any testing, we would have very few cases,” he said on May 14 during a speech at a medical supply distributor Owens & Minor facility in Allentown, Pennsylvania.
But presidential inconsistency doesn’t explain why key agencies like the CDC and the Food and Drug Administration badly bungled the testing rollout. In fact, they must shoulder a substantial share of the blame.
The CDC, weakened by nearly a decade of budget cuts, insisted on holding onto its bureaucratic power when it refused to use foreign tests or one authorized by the World Health Organization (WHO), which had been successfully deployed by Germany. The agency’s initial homegrown effort produced an inaccurate test, which set back the rollout by nearly a month.
The FDA, hemmed in by laws written to please a growing testing industry, has limited oversight over tests developed in private labs. As the pandemic spread, the declared emergency gave the agency new Emergency Use Authorization powers, which should have facilitated more rapid introduction of new tests into the market. Instead, the agency erected new roadblocks to getting a private, lab-developed test into the field. Then, as criticism mounted, it reversed course and waived dozens of new COVID-19 tests onto the market without providing data indicating their clinical efficacy.
In the wake of those flip-flops, some invalid tests began entering the market. Florida-based AdventHealth in mid-May announced it had to throw out the results of 35,000 tests from an outside lab it refused to name. That single lab at that point had conducted more than 5% of the state’s tests. Cleveland Clinic’s test of Abbott Labs’ ID NOW test, which gives results in 15 minutes and received praise at a White House briefing, missed 15% of cases when tested against known COVID-19 samples.
“That’s not too good,” Cleveland Clinic’s Dr. Gary Procop told NPR, suggesting a test should have a maximum of 5% false negatives. Hospitals, physicians and public health authorities that rely on tests with poor sensitivity (ones that identify fewer than 95% of actual cases) will have limited insight into the disease’s spread.
The problems may be in test administration. Current point-of-care tests require deep nasal swabs and newly approved at-home tests rely on shorter swabs that may not pick up pre-symptomatic or minor cases that can still infect other people.
The FDA also bungled the rollout of tests that measure SARS-CoV-2 antibodies in blood serum. Such tests, if accurate, would be extremely useful in determining who already had the disease without symptoms or treatment. Accurate serology tests would facilitate a more rapid reopening of the economy if the presence of antibodies conferred permanent or longer-term immunity. Unfortunately, that is still a big scientific unknown.
Action Steps for the U.S.
At a minimum, the U.S. must double the number of COVID-19 tests administered daily over the next month. There’s growing pressure to do even more. The Centers for Medicare and Medicaid Services in mid-May issued new guidelines for reopening nursing homes and assisted living complexes. It suggested operators make testing universal for residents and staff, which the trade association for the industry estimated would cost nearly a half billion dollars. As always, it did not make the requirements mandatory and left enforcement up to the states.
CMS also recommended these long-term care facilities show a declining caseload over a sustained period of time before allowing visitors. There are an estimated 1.5 million Americans living in such facilities, which, on average, employ about one care staff for every two residents.
Many workplaces eager to reopen will also need access to more testing before recalling their employees. It’s impossible to know how long it will take for the estimated 30 million Americans who’ve lost jobs to return to work. Many – like restaurants, grocery stores and entertainment venues – have public-facing jobs where employers will want to deploy frontend testing to reassure customers. The number of required tests could ramp up dramatically in the months ahead – well over the 4 to 5 million per week currently projected by the experts.
At the same time, the U.S. will need to ramp up its capacity to find and isolate people who’ve been exposed to people carrying the virus. Given the cash-strapped position of every state and local government, that activity will require a major investment by the federal government.
But more significantly, it will also take a major new commitment from the American people, since a successful tracing program will inevitably challenge some of their most cherished values.
Next: What it will take to implement a successful tracing program.